Goal
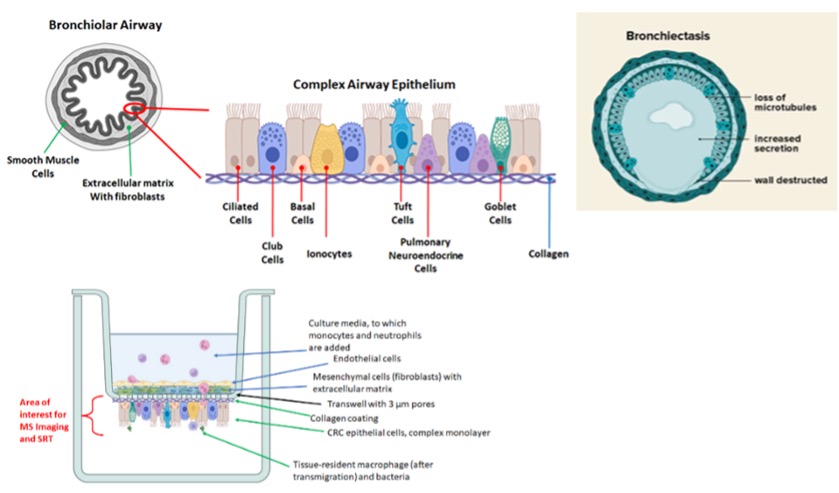
Most research in the CF lung considers the airway as a whole, likely averaging out all of the signals as if the whole lung were impacted equally and homogeneously; this undoubtedly leads to loss of information that is specific to the sites of infection and subsequent damage. New approaches must be applied to understand the focal nature of damage in the CF airway so that new therapies may be designed to target the changes in gene expression, protein function, and inflammatory processes that contribute to bronchiectasis (Fig.1). The biogeography of infection in the CF lung has been studied in two dimensions, across the epithelium, considering the topological relationship between two sites of infection by bacteria of the same or different species. We will extend biogeography into three dimensions by considering the relationship between those infection sites and the damage that occurs through the depth of the tissue, including in the mesenchyme, extracellular matrix, and endothelium. It is a well-accepted fact that it is not the bacteria that kills the CF patient, but the unrelenting cellular inflammatory response. The robust rate of transmigration of neutrophils and monocytes and release of proteases in the CF lung generates damage leading to bronchiectasis, induces epithelial-mesenchymal transition and fibrosis, and induces signaling pathways resulting in further inflammation. Airway-resident macrophages normally activate repair pathways, but these are defective in the CF lung.
This project seeks to build upon our recent successes that have led to deeper understanding of the earliest inciting events underlying lung damage in our youngest patients. This has given us the ability to recognize the earliest changes in the airway that underlie structural lung disease, evident by CT imaging. We propose to build upon this new understanding at the tissue level to identify molecular targets that may serve as the basis of new therapeutic development that will circumvent structural damage, in the lungs of our youngest patients, and perhaps even reverse the damage in the lungs of our older patients. This will require a multidisciplinary approach, from the following muti-investigator team based at Emory, Georgia Tech (GT), Georgia State, and the University of Alabama at Birmingham. [1-10]
Methods
Mass-spec imaging (metabolomics, proteomics), spatially resolved transcriptomics, barrier function assays, immunofluorescence, neutrophil transmigration, flow cytometry, untargeted metabolomics, MR Imaging of collagen deposition, animal models, imaging of bacteria in biofilms in situ.
Collaborators
- At Emory: Koval, Tirouvanziam, Kopp, Lama, Stecenko, Goldberg, Chandler, Guglani, Sueblinvong.
- At Georgia Tech: Takayama, Whiteley, Coskun, Garg, Diggle.
- At Georgia State: Azimi, Yang.
- At Univ. Alabama at Birmingham: Margaroli.
Funding
Children’s Healthcare of Atlanta, Inc. NIH proposal planned.
References
- Olivenca, D.V., J.D. Davis, C.M. Kumbale, C.Y. Zhao, S.P. Brown, N.A. McCarty, and E.O. Voit (2023) Mathematical models of cystic fibrosis as a systemic disease. WIREs Mech Dis. 15: e1625.
- Strickland, K.M., K. Neselu, A.J. Grant, C.L. Espy, N.A. McCarty, and I. Schmidt-Krey (2021) Reconstitution of detergent-solubilized membrane proteins into proteoliposomes and nanodiscs for functional and structural studies. Methods Mol Biol. 2302: 21-35.
- Eddins, D.J., J. Yang, A. Kosters, V.D. Giacalone, X. Pechuan-Jorge, J.D. Chandler, J. Eum, B.R. Babcock, B.S. Dobosh, M.R. Hernandez, F. Abdulkhader, G.L. Collins, D.Y. Orlova, R.P. Ramonell, I. Sanz, C. Moussion, F. Eun-Hyung Lee, R.M. Tirouvanziam, and E.E.B. Ghosn (2023) Transcriptional reprogramming of infiltrating neutrophils drives lung pathology in severe covid-19 despite low viral load. Blood Adv. 7: 778-799.
- Viola, H., K. Washington, C. Selva, J. Grunwell, R. Tirouvanziam, and S. Takayama (2022) A high-throughput distal lung air-blood barrier model enabled by density-driven underside epithelium seeding. Adv Healthc Mater. 11: e2102450.
- Dobosh, B., V.D. Giacalone, C. Margaroli, and R. Tirouvanziam (2021) Mass production of human airway-like neutrophils via transmigration in an organotypic model of human airways. STAR Protoc. 2: 100892.
- Forrest, O.A., S.A. Ingersoll, M.K. Preininger, J. Laval, D.H. Limoli, M.R. Brown, F.E. Lee, B. Bedi, R.T. Sadikot, J.B. Goldberg, V. Tangpricha, A. Gaggar, and R. Tirouvanziam (2018) Frontline science: Pathological conditioning of human neutrophils recruited to the airway milieu in cystic fibrosis. J Leukoc Biol. 104: 665-675.
- Hartl, D., R. Tirouvanziam, J. Laval, C.M. Greene, D. Habiel, L. Sharma, A.O. Yildirim, C.S. Dela Cruz, and C.M. Hogaboam (2018) Innate immunity of the lung: From basic mechanisms to translational medicine. J Innate Immun. 10: 487-501.
- Cai, S., T. Hu, M. Venkatesan, M. Allam, F. Schneider, S.S. Ramalingam, S.Y. Sun, and A.F. Coskun (2022) Multiplexed protein profiling reveals spatial subcellular signaling networks. iScience. 25: 104980.
- Ganesh, S., T. Hu, E. Woods, M. Allam, S. Cai, W. Henderson, and A.F. Coskun (2021) Spatially resolved 3d metabolomic profiling in tissues. Sci Adv. 7: eabd0957.
- Margaroli, C., T. Fram, N.S. Sharma, S.B. Patel, J. Tipper, S.W. Robison, D.W. Russell, S.D. Fortmann, M.M. Banday, Y. Soto-Vazquez, T. Abdalla, S. Saitornuang, M.C. Madison, S.M. Leal, Jr., K.S. Harrod, N.B. Erdmann, and A. Gaggar (2023) Interferon-dependent signaling is critical for viral clearance in airway neutrophils. JCI Insight. 8: e167042.

