Goal
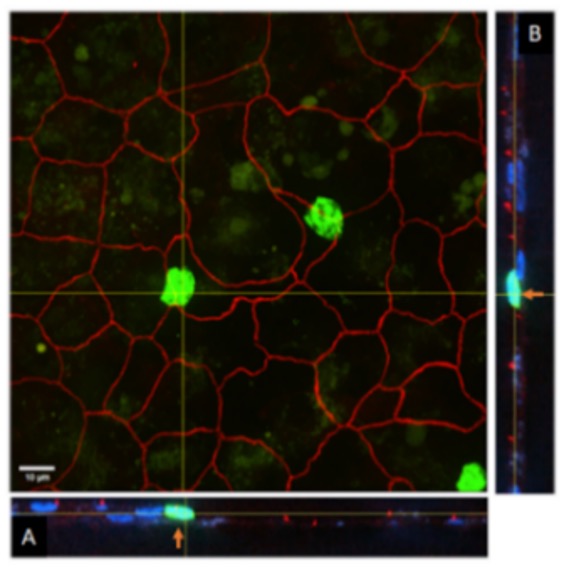
CF is the second most common lethal genetic disease among Americans, arising from mutations in the gene encoding CFTR. CF-related diabetes (CFRD), the most common co-morbidity[1], begins to develop early in childhood and the pathological consequences are severe including significantly increased frequency of acute pulmonary exacerbations and increased rate of decline in lung function compared to CF without diabetes, resulting in poorer outcomes[2-4]. Recent [5] and prior work in the McCarty lab and Koval lab, and others, identified multiple defects in CF airway epithelial cells related to glucose handling [6] and expression and function of tight junction (TJ) proteins [7] that are responsible for the barrier function of the airway monolayer. We most recently showed that expression of mutant CFTR in airway epithelial cells strongly impacts the localization of multiple important TJ proteins, that this is worsened under conditions of hyperglycemia, and only partly recovered by treatment with CFTR correctors (see below) [5]. New preliminary data show that the actin cytoskeleton is disrupted in CF cells, which can alter TJ protein assembly and function, consistent with previous reports [8, 9]. However, these prior studies do not address the impact of impaired TJ function or actin cytoskeleton disruption on polymorphonuclear neutrophils (PMNs) that are recruited to the CF airway in response to muco-obstruction and infection. It is the damage produced by PMNs that leads to most of the morbidity and mortality in CF.
To enter the airway in response to pathogens, PMNs must exit the vasculature, cross the interstitium, and migrate across the airway epithelium (Fig. 1). However, mechanistic studies of how mutant CFTR in airway epithelial cells controls the rate and extent of PMN Trans-Epithelial Migration (TEpM) and impacts PMN function are lacking. The Tirouvanziam lab identified a new paradigm for how PMNs turn pathogenic early on upon exposure to the CF airway milieu (the “GRIM” fate) [10-16]. Notably, on their way to the lumen, PMNs release small amounts of proteases that cause cumulative, progressive destruction along their migratory path in the lamina propria [17-19], which ultimately causes bronchiectasis [19] [20-23]. Our data show that PMNs cause worse damage during TEpM across primary CF airway monolayers than non-CF controls. Hence, the rate of TEpM and activity in the sub-apical space are likely to strongly induce lung injury in CF, leading to loss of lung function and death.
The overall goal of this project is to identify the mechanisms regulating interactions between PMNs and the airway epithelium [11] that occur during TEpM, how these are altered in the CF airway, and how they are further altered under conditions mimicking CFRD. We hypothesize that the loss of CFTR in the apical plasma membrane of airway epithelial cells destabilizes the actin-dependent TJ assembly complex, leading to shifts in the association of specific TJ proteins with the plasma membrane at sites of cell-cell contact, and that this induces alterations in interactions between transmigrating PMNs and epithelial cells themselves. We also hypothesize that cell-cell interactions are further defective in the context of hyperglycemic conditioning of the epithelium, and that this leads to both retention of PMNs in the sub-apical compartment and their partial activation to adopt the GRIM phenotype, thereby leading to greater destruction of the airway epithelium. [24-27]
Methods
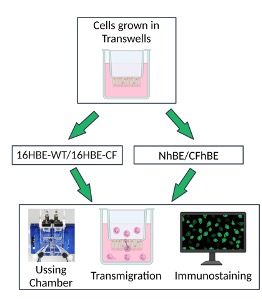
Flow cytometry, gene expression (RNA-Seq in both epithelial cells and neutrophils), transepithelial current measurements, immunofluorescence, neutrophil transmigration assays, bacterial killing assays, metabolic activity assays, novel assay for impact of neutrophil transmigration on airway barrier function, spatial transcriptomics, spatial metabolomics (Fig. 2).
Collaborators
Rabin Tirouvanziam, PhD, Dept. of Pediatrics, Emory. Mike Koval, PhD, Dept. of Medicine, Emory.
Funding
CF Foundation, grant MCCART21G0; CFF grant under review; NIH R01 planned
References
- Putman, M.S., A.W. Norris, R.L. Hull, M.R. Rickels, L. Sussel, S.M. Blackman, C. L. Chan, K.L. Ode, T. Daley, A.A. Stecenko, A. Moran, M.J. Helmick, S. Cray, J.A. Alvarez, V.A. Stallings, K.L. Tuggle, J.P. Clancy, T.L. Eggerman, J.F. Engelhardt, and A. Kelly (2023) Cystic fibrosis-related diabetes workshop: Research priorities spanning disease pathophysiology, diagnosis, and outcomes. Diabetes.
- Stecenko, A.A. and A. Moran (2010) Update on cystic fibrosis-related diabetes. Current opinion in pulmonary medicine. 16: 611-5.
- van den Berg, J.M., J.M. Kouwenberg, and H.G. Heijerman (2009) Demographics of glucose metabolism in cystic fibrosis. Journal of cystic fibrosis : official journal of the European Cystic Fibrosis Society. 8: 276-9.
- Kelsey, R., F.N. Manderson Koivula, N.H. McClenaghan, and C. Kelly (2019) Cystic fibrosis-related diabetes: Pathophysiology and therapeutic challenges. Clin Med Insights Endocrinol Diabetes. 12: 1179551419851770.
- Vazquez Cegla, A.J., K.T. Jones, G. Cui, K.A. Cottrill, M. Koval, and N.A. McCarty (2024) Effects of hyperglycemia on airway epithelial barrier function in wt and cf 16hbe cells. Sci Rep. 14: 25095.
- Molina, S.A., H.K. Moriarty, D.T. Infield, B.R. Imhoff, A.H. Kim, J.M. Hansen, W.R. Hunt, M. Koval, and N.A. McCarty (2017) Insulin signaling via akt2 regulates the airway glucose barrier in a cftr-dependent manner. Am. J. Physiol.: Lung Cell Mol Physiol. 312: L688-L702.
- Molina, S.A., B. Stauffer, H.K. Moriarty, A.H. Kim, N.A. McCarty, and M. Koval (2015) Junctional abnormalities in human airway epithelial cells expressing f508del cftr. American journal of physiology. Lung cellular and molecular physiology. 309: L475-87.
- Castellani, S., M. Favia, L. Guerra, A. Carbone, A.C. Abbattiscianni, S. Di Gioia, V. Casavola, and M. Conese (2017) Emerging relationship between cftr, actin and tight junction organization in cystic fibrosis airway epithelium. Histol Histopathol. 32: 445-459.
- Castellani, S., L. Guerra, M. Favia, S. Di Gioia, V. Casavola, and M. Conese (2012) Nherf1 and cftr restore tight junction organisation and function in cystic fibrosis airway epithelial cells: Role of ezrin and the rhoa/rock pathway. Lab Invest. 92: 1527-40.
- Tirouvanziam, R., I. Khazaal, and B. Peault (2002) Primary inflammation in human cystic fibrosis small airways. American journal of physiology. Lung cellular and molecular physiology. 283: L445-51.
- Tirouvanziam, R., Y. Gernez, C.K. Conrad, R.B. Moss, I. Schrijver, C.E. Dunn, Z.A. Davies, L.A. Herzenberg, and L.A. Herzenberg (2008) Profound functional and signaling changes in viable inflammatory neutrophils homing to cystic fibrosis airways. Proceedings of the National Academy of Sciences of the United States of America. 105: 4335-9.
- Makam, M., D. Diaz, J. Laval, Y. Gernez, C.K. Conrad, C.E. Dunn, Z.A. Davies, R.B. Moss, L.A. Herzenberg, L.A. Herzenberg, and R. Tirouvanziam (2009) Activation of critical, host-induced, metabolic and stress pathways marks neutrophil entry into cystic fibrosis lungs. Proceedings of the National Academy of Sciences of the United States of America. 106: 5779-83.
- Laval, J., J. Touhami, L.A. Herzenberg, C. Conrad, N. Taylor, J.L. Battini, M. Sitbon, and R. Tirouvanziam (2013) Metabolic adaptation of neutrophils in cystic fibrosis airways involves distinct shifts in nutrient transporter expression. J Immunol. 190: 6043-50.
- Ingersoll, S.A., J. Laval, O.A. Forrest, M. Preininger, M.R. Brown, D. Arafat, G. Gibson, V. Tangpricha, and R. Tirouvanziam (2015) Mature cystic fibrosis airway neutrophils suppress t cell function: Evidence for a role of arginase 1 but not programmed death-ligand 1. J Immunol. 194: 5520-8.
- Forrest, O.A., S.A. Ingersoll, M.K. Preininger, J. Laval, D.H. Limoli, M.R. Brown, F.E. Lee, B. Bedi, R.T. Sadikot, J.B. Goldberg, V. Tangpricha, A. Gaggar, and R. Tirouvanziam (2018) Frontline science: Pathological conditioning of human neutrophils recruited to the airway milieu in cystic fibrosis. J Leukoc Biol. 104: 665-675.
- Margaroli, C., D. Moncada-Giraldo, D.A. Gulick, B. Dobosh, V.D. Giacalone, O.A. Forrest, F. Sun, C. Gu, A. Gaggar, H. Kissick, R. Wu, G. Gibson, and R. Tirouvanziam (2021) Transcriptional firing represses bactericidal activity in cystic fibrosis airway neutrophils. Cell Rep Med. 2: 100239.
- Liou, T.G. and E.J. Campbell (1996) Quantum proteolysis resulting from release of single granules by human neutrophils: A novel, nonoxidative mechanism of extracellular proteolytic activity. J Immunol. 157: 2624-31.
- Zay, K., S. Loo, C. Xie, D.V. Devine, J. Wright, and A. Churg (1999) Role of neutrophils and alpha1-antitrypsin in coal- and silica-induced connective tissue breakdown. The American journal of physiology. 276: L269-79.
- Zen, K., Y.L. Guo, L.M. Li, Z. Bian, C.Y. Zhang, and Y. Liu (2011) Cleavage of the cd11b extracellular domain by the leukocyte serprocidins is critical for neutrophil detachment during chemotaxis. Blood. 117: 4885-94.
- Margaroli, C. and R. Tirouvanziam (2016) Neutrophil plasticity enables the development of pathological microenvironments: Implications for cystic fibrosis airway disease. Mol Cell Pediatr. 3: 38.
- Margaroli, C., L.W. Garratt, H. Horati, A.S. Dittrich, T. Rosenow, S.T. Montgomery, D.L. Frey, M.R. Brown, C. Schultz, L. Guglani, A. Kicic, L. Peng, B.J. Scholte, M.A. Mall, H.M. Janssens, S.M. Stick, and R. Tirouvanziam (2019) Elastase exocytosis by airway neutrophils is associated with early lung damage in children with cystic fibrosis. American journal of respiratory and critical care medicine. 199: 873-881.
- Genschmer, K.R., D.W. Russell, C. Lal, T. Szul, P.E. Bratcher, B.D. Noerager, M. Abdul Roda, X. Xu, G. Rezonzew, L. Viera, B.S. Dobosh, C. Margaroli, T.H. Abdalla, R.W. King, C.M. McNicholas, J.M. Wells, M.T. Dransfield, R. Tirouvanziam, A. Gaggar, and J.E. Blalock (2019) Activated pmn exosomes: Pathogenic entities causing matrix destruction and disease in the lung. Cell. 176: 113-126 e15.
- Giacalone, V.D., C. Margaroli, M.A. Mall, and R. Tirouvanziam (2020) Neutrophil adaptations upon recruitment to the lung: New concepts and implications for homeostasis and disease. Int J Mol Sci. 21.
- Olivenca, D.V., D. J.D., N.A. McCarty, R.R. Pinto, and E.O. Voit, The system of cystic fibrosis., in Systems medicine: Integrative, qualitative and computational approaches, O. Wolkenhauer, Editor. 2021. p. 295-305.
- Olivenca, D.V., J.D. Davis, C.M. Kumbale, C.Y. Zhao, S.P. Brown, N.A. McCarty, and E.O. Voit (2023) Mathematical models of cystic fibrosis as a systemic disease. WIREs Mech Dis. 15: e1625.
- Cottrill, K.A., S.T. Stephenson, A.F. Mohammad, S.O. Kim, N.A. McCarty, R. Kamaleswaran, A.M. Fitzpatrick, and J.D. Chandler (2023) Exacerbation-prone pediatric asthma is associated with arginine, lysine, and methionine pathway alterations. J Allergy Clin Immunol. 151: 118-127 e10.
- McCarty, N.A. (2021) Breathe - your immune system is counting on it. The Journal of experimental medicine. 218.

