Goal
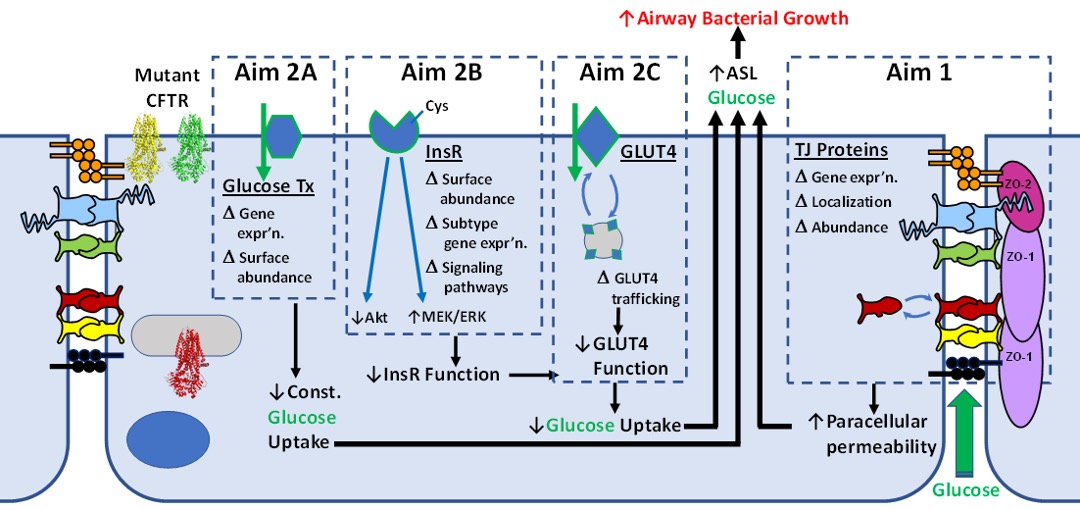
Mutations in the gene encoding the CFTR Cl- channel protein underlie Cystic Fibrosis (CF), one of the most common lethal genetic diseases in the U.S. Loss of CFTR function leads to the well-known triad of infection, inflammation, and obstruction that result in progressive loss of lung function [1]. These pulmonary complications are amplified as patients develop CF-related diabetes (CFRD), the most common comorbidity. The pathological consequences of diabetes in CF patients are severe: CFRD patients have a significant increase in the frequency of pulmonary exacerbations which correlates with an accelerated decline in lung function that dramatically shortens lifespan. Little is known about how the inability to control circulating glucose levels impacts airway function in CF, leading to more rapid decline. The goals of this project are to identify the mechanistic link(s) between dysfunction of CFTR and worsening of pulmonary disease associated with hyperglycemia and CFRD (Fig.1). We focus on the linked questions of how airway epithelial cells and airway-resident bacteria adapt to systemic hyperglycemia, why that process is defective in CF, and how this impacts the clinically relevant function of clearance of infection from the lung. We also focus on the signaling pathways between the insulin receptor and regulation of tight junction function, which are curiously disrupted in cells expressing mutant CFTR. [1-11]
Methods
Gene expression (qRT-PCR and RNA-Seq), immunofluorescence, glucose transporter translocation, transepithelial current measurements, development of automated cell culture system, modeling of continuous glucose monitoring data in people with CFRD, co-culture of epithelial cells with bacteria, glucose uptake assays, redox assays, cell signaling pathways.
Collaborators
Mike Koval, PhD, Dept. of Medicine, Emory; Joshua Chandler, PhD, Dept. of Pediatrics, Emory; Tanicia Daley Jean Pierre, MD, Dept. of Pediatrics, Emory; Shilin Zhao, PhD, Vanderbilt University.
Funding
NIH grant 1R01HL176542; CF Foundation grant MCCART24G0; NIH P30 DK125013.
References
- Olivenca, D.V., J.D. Davis, C.M. Kumbale, C.Y. Zhao, S.P. Brown, N.A. McCarty, and E.O. Voit (2023) Mathematical models of cystic fibrosis as a systemic disease. WIREs Mech Dis. 15: e1625.
- Molina, S.A., H.K. Moriarty, D.T. Infield, B.R. Imhoff, R.J. Vance, A.H. Kim, J.M. Hansen, W.R. Hunt, M. Koval, and N.A. McCarty (2017) Insulin signaling via the pi3-kinase/akt pathway regulates airway glucose uptake and barrier function in a cftr-dependent manner. American journal of physiology. Lung cellular and molecular physiology. 312: L688-L702.
- (1986) Social security programs in the united states. Soc Secur Bull. 49: 5-59.
- Molina, S.A., B. Stauffer, H.K. Moriarty, A.H. Kim, N.A. McCarty, and M. Koval (2015) Junctional abnormalities in human airway epithelial cells expressing f508del cftr. American journal of physiology. Lung cellular and molecular physiology. 309: L475-87.
- Hunt, W.R., S.M. Zughaier, D.E. Guentert, M.A. Shenep, M. Koval, N.A. McCarty, and J.M. Hansen (2014) Hyperglycemia impedes lung bacterial clearance in a murine model of cystic fibrosis-related diabetes. American journal of physiology. Lung cellular and molecular physiology. 306: L43-9.
- Monge, M.E., J.J. Perez, P. Dwivedi, M. Zhou, N.A. McCarty, A.A. Stecenko, and F.M. Fernandez (2013) Ion mobility and liquid chromatography/mass spectrometry strategies for exhaled breath condensate glucose quantitation in cystic fibrosis studies. Rapid Commun Mass Spectrom. 27: 2263-71.
- Maliniak, M.L., A.A. Stecenko, and N.A. McCarty (2016) A longitudinal analysis of chronic mrsa and pseudomonas aeruginosa co-infection in cystic fibrosis: A single-center study. Journal of cystic fibrosis : official journal of the European Cystic Fibrosis Society. 15: 350-6.
- Zughaier, S.M., B.B. Stauffer, and N.A. McCarty (2014) Inflammation and er stress downregulate bdh2 expression and dysregulate intracellular iron in macrophages. J Immunol Res. 2014: 140728.
- Vazquez Cegla, A.J., C. Hedden, B.R. Imhoff, G. Cui, and N.A. McCarty (2024) Development of a programmable automated cell culture system to study the lung pathophysiology of cystic fibrosis-related diabetes. Heliyon. 10: e37977.
- Cui, G., D.A. Moustafa, S. Zhao, A.V. Cegla, J.T. Lyles, J.B. Goldberg, J.D. Chandler, and N.A. McCarty (2024) Chronic hyperglycemia aggravates lung function in a scnn1b-tg murine model. American journal of physiology. Lung cellular and molecular physiology. 327: L473-L486.
- Vazquez Cegla, A.J., K.T. Jones, G. Cui, K.A. Cottrill, M. Koval, and N.A. McCarty (2024) Effects of hyperglycemia on airway epithelial barrier function in wt and cf 16hbe cells. Sci Rep. 14: 25095.

