Our Research
We are interested in how transcription and chromatin regulation facilitate cellular "decision-making", allowing stem cells and mature cells to adopt and maintain different functional states.
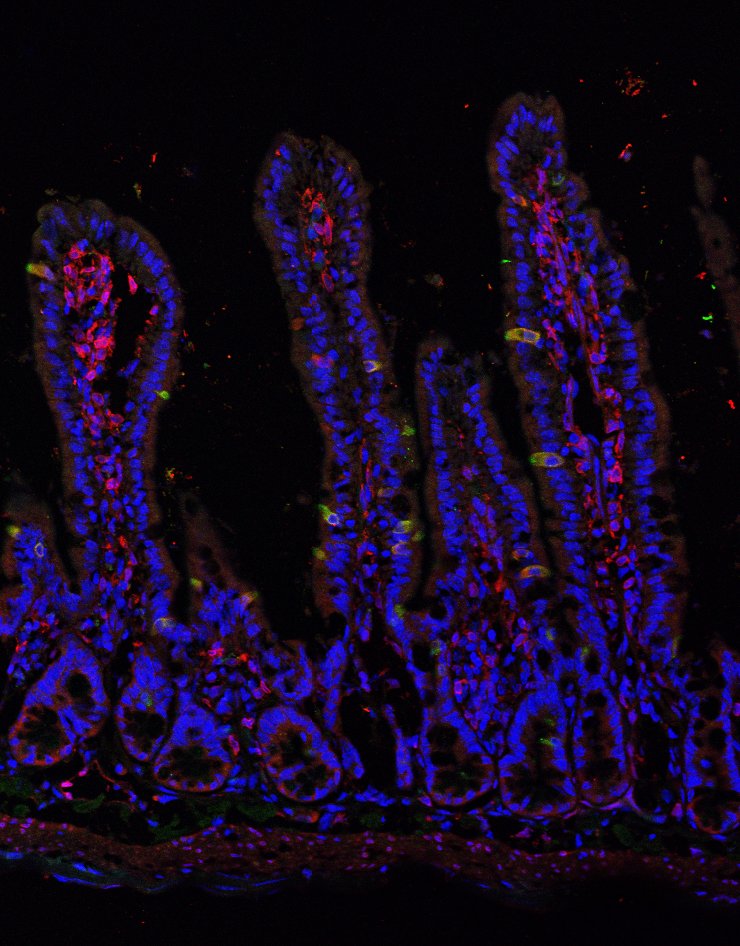
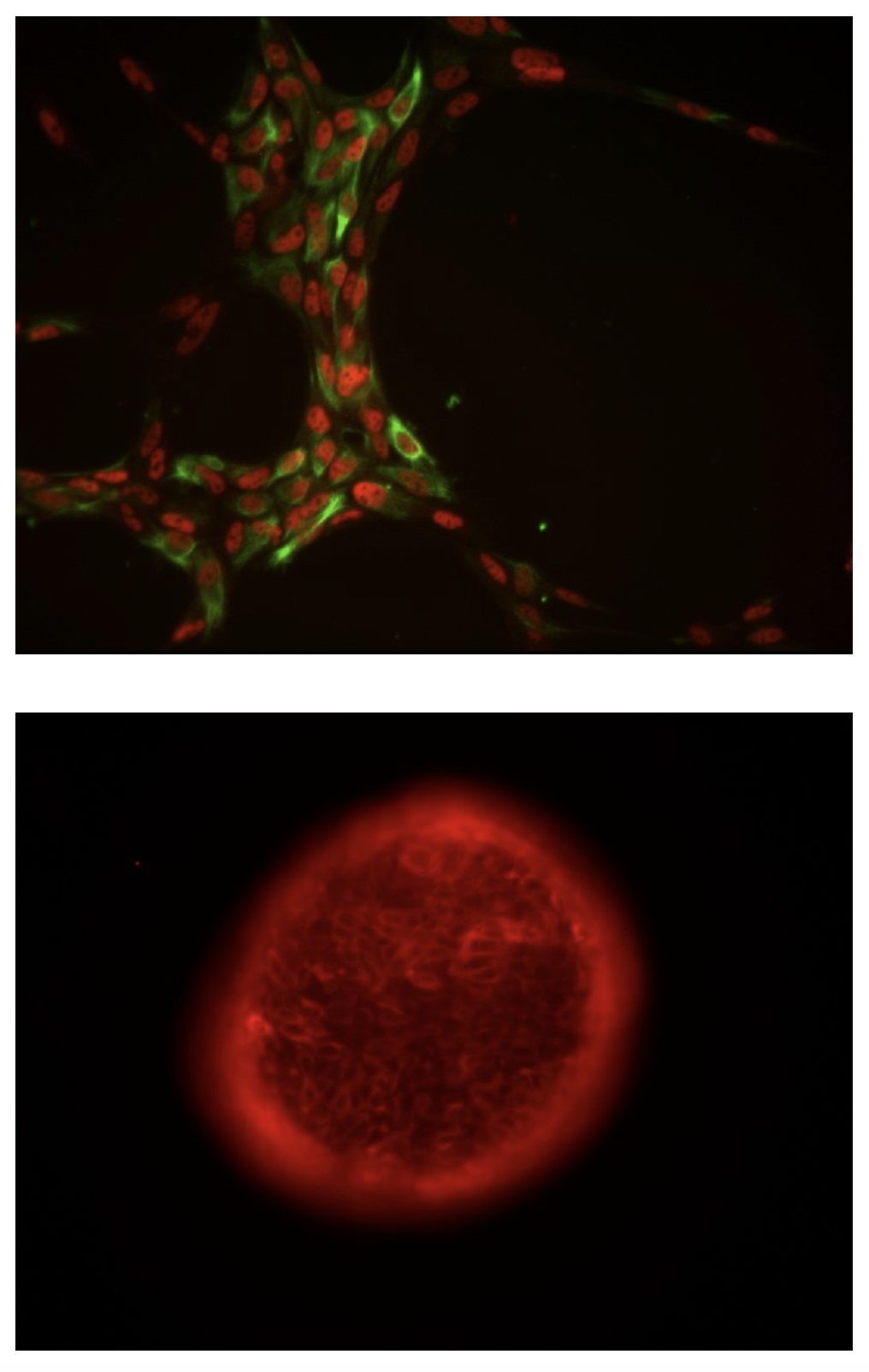
Chromatin regulation in intestinal stem cell biology
Proliferating adult tissues are maintained by somatic stem cells, which produce all of a tissue’s differentiated cells under normal conditions, initiate regeneration following injury, and are thought to drive tumorigenesis after acquiring oncogenic mutations. The intestinal epithelium exhibits significant stem cell activity via constant, rapid renewal throughout adult life. In fact, cells in the intestinal epithelium undergo near-total renewal every 5-7 days; during which time intestinal stem cells (ISCs) drive proliferation and differentiation along a crypt/villus axis. Progenitor cells can "de-differentiate" to acquire ISC characteristics and function in scenarios where normal ISCs become compromised, effectively forming two stem cell pools: "active" ISCs and progenitor cells that function as "reserve" or "facultative" ISCs. These characteristics place high demands on the cellular potential of ISC populations: how do these cells maintain normal function, make differentiation decisions, and retain the ability to revert to a less differentiated state under stress or damage?
To address these questions, we're studying how chromatin affects ISC potential to make reversible and irreversible differentiation and self-renewal decisions. Chromatin refers to the complement of post-translational modifications to DNA-associated histone proteins as well as modifications to DNA itself. Together, these epigenetic modifications (or "marks") have the potential to impact gene expression and cellular behavior on a global scale. Our studies are aimed at mapping these marks and determining their functional and mechanistic significance. By understanding global "epigenomic" regulation of ISCs, we hope to better understand how these regulatory networks change in the setting of injury, inflammatory disease, and cancer.

Regeneration and cell fate in the intrahepatic bile ducts
The liver is key regulator of organismal homeostasis and is well known for its remarkable ability to regenerate following physical injury or chemically induced damage. However, evidence suggests that all epithelial regeneration in the adult liver is drive by mature hepatocytes and biliary epithelial cells (BECs) rather than a dedicated stem cell pool. In cases of chronic or severe injury, cells can transdifferentiate between hepatocyte and BEC lineages. We are pursuing a deeper understanding of transcriptional and chromatin regulation of BECs during homeostasis and following injury. We are particularly interested in the mechanisms regulating BEC proliferation and transdifferentiation during ductular reaction, an extensive remodeling and expansion of the intrahepatic bile ducts that occurs in acute and chronic liver injury.
We are also applying tools and techniques developed in our studies of intestinal stem cells (ISCs) to understand BEC heterogeneity. Our long-term goal is to better understand functional and genomic heterogeneity among BECs to advance our understanding of hepatobiliary disease to inform the development of novel therapeutics.